|
#"2 e"^(-)"/ orbital" xx "5 orbitals" = "10 e"^(-)#Įach of these ten electrons will have its unique set of four quantum numbers. 
3 the partial DOS (PDOS) projected onto the five 4d orbitals of the Ru ion in the RuO 6 octahedral local coordinates (xyz. 
Now, since each orbital can hold a maximum of two electrons, one with spin-up and one with spin-down, it follows that the d-obitals can hold a total of To identify the orbital ordering state in Sr 2 RuO 4, we present in Fig. #m_l = #Įach of these five values describes one of the five d-orbitals available in a d-subshell.įinally ,the spin quantum number, #m_s#, can only take two values, #-1/2# for an electron that has spin-down and #+1/2# for an electron that has spin-up. For any d-subshell, the magnetic quantum number can take the values The specific orbital in which the electron is located is given by the magnetic quantum number, #m_l#. Since you're looking for the d-subshell, you will need #l=2#. A wave function for an electron in an atom is called an atomic orbital this atomic. The subshell in which the electron is located is described by the angular magnetic quantum number, #l#, which for the fourth energy level takes the following values #n = color(red)(4) -># the electron is located on the fourth energy level So, the principal quantum number, #n#, describes the energy level on which the electron is located. Now, you are given a #color(red)(4)d# orbital and asked to find how many sets of quantum numbers can describe an electron located in such an orbital, or, in other words, how many electrons can occupy a #color(red)(4)d# orbital. This 4d orbital is one of four four-lobed orbitals (called 4dxy, 4dyz, 4dxz and 4dx2-y2) arranged around the atom. 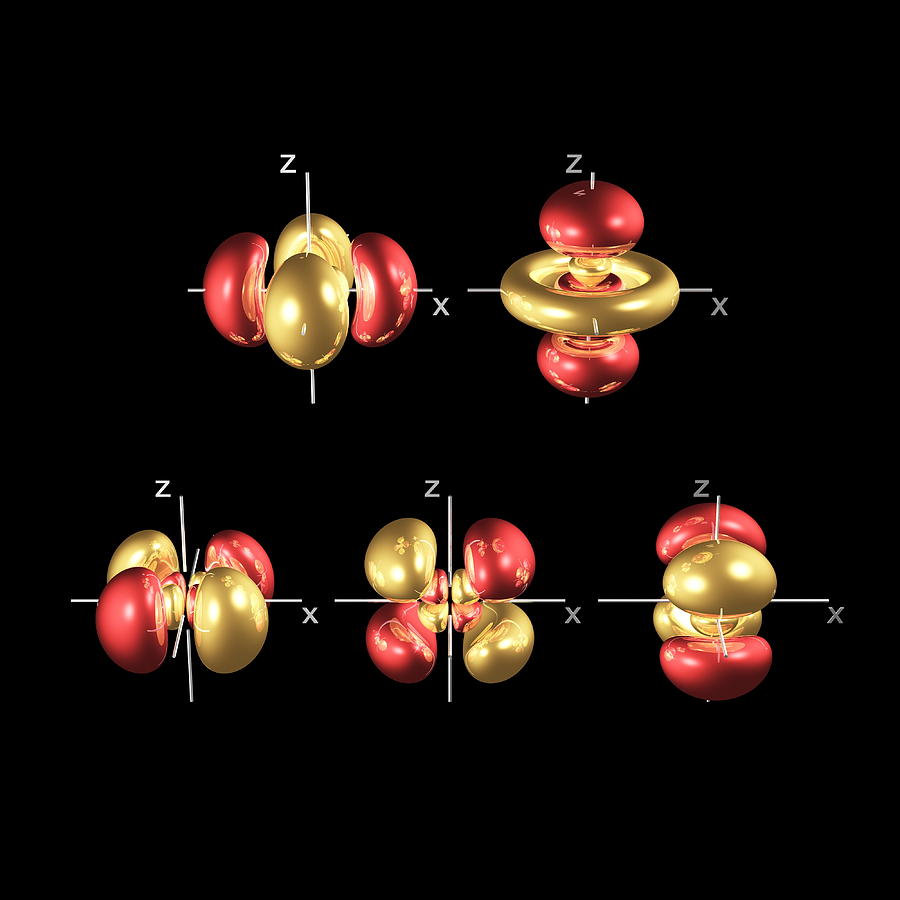
so thats 5 orbitals, and 2 electrons can fit into each one (one with a +1/2 spin and one with a. As you know, we use four quantum numbers to describe the position and spin of an electron in an atom.Įach electron has its unique set of quantum numbers, which means that two electrons can share one, two, or even three quantum numbers, but never all four. because a d subshell corresponds to an l value of 2, ml can be -2,-1,0,1,2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
